Baking Soda and Vinegar Lab
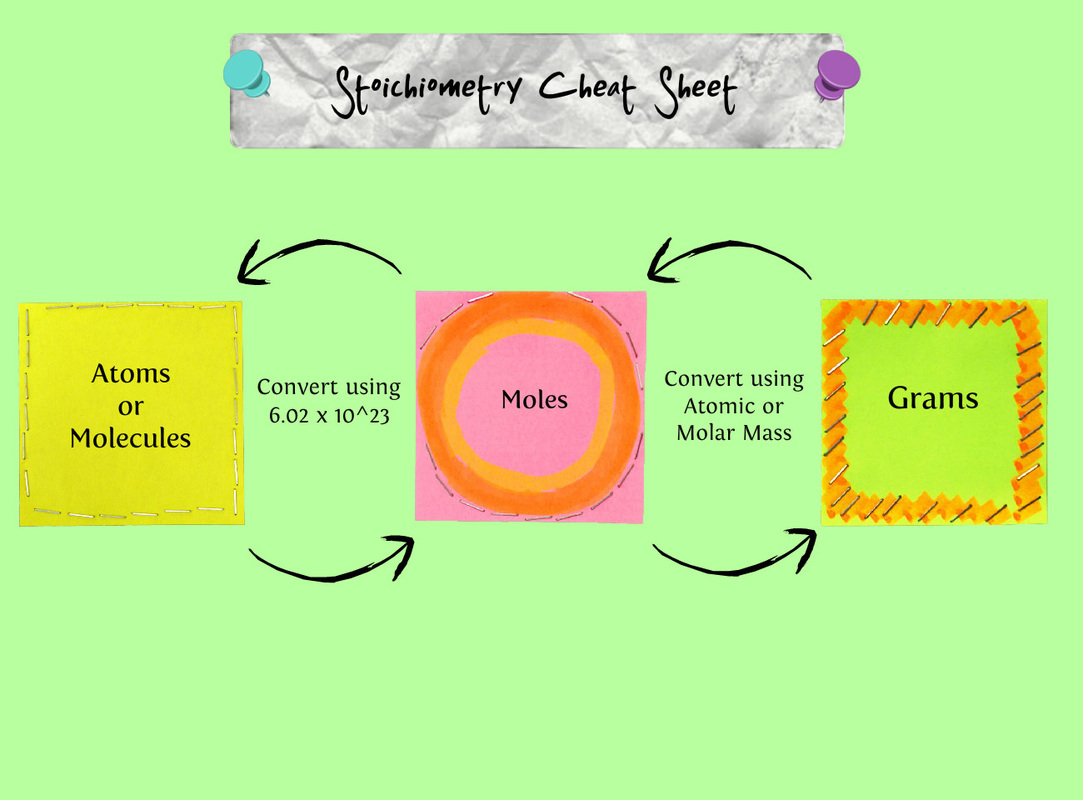
I think over the course of my life I have done this lab plenty of times. I just never realized that it related to Chemistry, let alone a chemical reaction that you can turn into a formula using the Periodic Table.The Baking Soda and Vinegar lab is the old "volcano reaction " lab, or at least that's what I thought when watching the reaction happening.To explain what is happening: The Baking soda is reacting with the vinegar and creates CO2 gases, like the kind we exhale and that cars produce. This is also what causes the fizzy taste in Cola and other sodas. There are all kinds of other experiments; one blows up a balloon, another blows up bottles. I really don't have much else to say because of the simplicity of this lab. Along with the Baking Soda and Vinegar Lab Post I will also be talking about Moles. What is a mole? A chemical mole is a unit of measure, just like a gram or an ounce. It is used internationally so that all chemists speak the same measurement language. The mole was invented because, well, it made sense. Scientists were having a hard time converting between atoms of an element and grams of an element so scientists came up with a "mole." Avogadro's # You might recognize 6.02x1023 as Avogadro’s number; this number is used as a constant throughout chemistry, and here I'm going to use it to define the mole. Usually, moles refer to particles that make up a certain amount of an element, and we use moles to measure how much of a substance is reacting in a chemical equation. However, you can also measure other things in moles. Example: A mole of horses would be 6.02x1023 horses . . . which is actually quite a lot. When you think about a mole as 602,000,000,000,000,000,000,000 horses, it seems like way too big of a number to be describing something that fits in a beaker in the chem lab! However, because atoms are so small there are bunches of atoms in everything we’re measuring. Therefore, a mole is actually a very appropriate way to measure chemical substances. Another benefit of using moles to measure substances is that it directly correlates to the number of atoms and molecules and grams. A mole tells you what fractional part of Avogadro’s number you’re working with. Example: If you have .25 mol, you would have .25 (or 25%) of Avogadro’s number, which is 1.505x1023 (.25 * 6.02x1023=1.505x1023). How do you convert Moles and Grams? Converting from grams to moles is really quite easy. The first thing you need is a Periodic Table. Then, read over the specific problem you want to complete and decide if you need to convert from grams to moles, or from moles to grams. After you’ve identified the problem and the conversion, find the element you are working with on the periodic table. Then, locate the atomic weight. If you are converting an amount in grams to moles, you will be dividing by the atomic weight. If you are converting an amount in moles to grams, you will be multiplying by the atomic weight, but if you use dimensional analysis you will not have to worry about figuring out the operations before you do the problem. Dimensional analysis is similar to setting up a table and it is waaaaayyy easier to understand.
0 Comments
Stoichiometry is all about the amount of substances that are involved in a chemical reaction. You can use stoichiometry to find the amount of a substance before a reaction, or even how much of a material that is produced after the reaction.
Reactions are dependent on the amount of product you have. So stoichiometry can help you figure out how much of a compound you will need, or how much you should start off with. Almost all of there equations can be solved in 4 steps, and at first it may seen hard, but after practice it becomes easier. 1.Balance the equation. 2. Convert units of a given substance to moles. 3. Using the mole ratio, calculate the moles of substance yielded by the reaction. 4. Convert moles of wanted substance to desired units. #1 20 Ml of Distilled Water
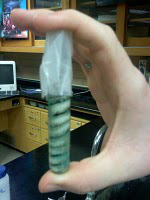
2 Grams CuCl2 1.6 Grams Aluminum #2 20 Ml of Distilled Water 4 Grams CuCl2 1.8 Grams Aluminum #3 30 Ml of Distilled Water 2 Grams CuCl2 2.2 Grams Aluminum The green color is the experiment is the copper ions reacting, which then created the maroon color at the end of the lab. The maroon copper is the aluminum being displaced by the copper. The less aluminum to copper chloride will create a less intense reaction. Distilled water - The tap water will change the reaction, but you can use either tap or distilled water. You can even do the same lab twice with both types of water and compare. What eventually happens when you put Aluminum and Copper Chloride together? When you put aluminum in copper chloride, the copper together the chloride eats away at the aluminum. There is a slight burning smell and smoke as a result of the chemical reaction. As the copper chlorides works away at the aluminum, the aluminum turning into a darkish maroon color. There are all kinds of Chemical Reactions that happen around us all the time. By being able to identify and classify these reactions helps us to make sense of them and we are then able to predict the product. In this lab involving Silver and Copper I was with a group of two others. The whole end product of the lab was to make silver metal. This lab was separated into a three day time span in order to make our own silver. Copper wire reacts with aqueous silver nitrate. The relative amounts (moles) of reactant and product are determined from the mass loss of the copper wire, the starting mass of the silver nitrate, and the mass of the silver metal obtained. During this lab the copper will be changed from its elemental form to its aqueous blue ion form. At the same time, silver ions will be removed from the solution and will be deposited on the wire in the elemental Ag metallic form. Day 1: On Day 1 we cut 30 cm worth of copper wire and then dinner it down with steel wool. And them we coiled the wire around the pencil forming a loose coil. Then we stretched the site so it would be able to for inside of the tube. We then made sure to measure everything used in the lab. Including the coil, and the dish of silver nitrate. Which we all used the specific amount of 1.0 gram. After we poured the silver nitrate into the test tube with the copper coil we put distilled water until the water was 2 cm from the top. And then we covered the top with parafilm ( wax paper on steroids, its actually pretty fun and cool to mess with.) We then waited till Day 2 to continue with the lab. Day 2: For day two we took a piece of filter paper and used it to separate the silver. And then we shook the test tube to separate the copper wire from the silver. Them we set up a funnel with the filter paper. With a waste beaker under the funnel we carefully lifted the copper wire out of the test tube and held it over our magical filter system. Then we washed the silver off with water and it ran down the wire onto the filter. After we did this we wanted the copper to dry so we put it on a piece of paper. Then we separated the silver. Then you have to wait for it to dry overnight. Day 3: Day three was the easiest out of them all. All we had to do was weigh the copper coil and record its mass. And also weigh the silver and filter paper and then record it's mass. Mass of silver nitrate: 1.0 gram Mass of copper coil before reaction: 3.44 grams Mass of copper coil after reaction: 3.252 grams Mass of filter paper & silver: 2.165 grams Mass of filter paper: 1.52 grams Mass of silver produced: .645 grams The correct equation for the Chemical Reaction is 2AgNO3 + Cu --> Cu(NO3)2 + 2Ag How close were your actual results to the results that you calculated? gAg .645 grams gCu .188 grams After about fifteen minutes after we placed the copper in the water with the silver nitrate this is what it looked like.
What is Dimensional Analysis?
Sound scary. Well it's not all that bad. Dimensional Analysis is a mathematical system using conversion factors to move from one unit of measurement to a different unit of measurement. For example, if you need to calculate how many seconds are in a day, you can use dimensional analysis to set all of your needed conversions into one problem. Dimensional Analysis is both very helpful in Science and as well as in Math. The work I have shown below is some notes and examples I did during class in Chem. When do you use Dimensional Analysis in Chemistry? Conversion factors and dimensional analysis make comparing different units much easier. These conversions are necessary in many calculations in Chemistry. Equations in physics problems may require calculations using derived units. Derived units are those that contain two separate measurement units that are used as one. Example: The unit for force is a newton (N). A newton is equal to 1 kilogram x meter/second . Calculations in chemistry require comparisons of measurements of material in solid, liquid, and gas form. Dimensional analysis makes these comparisons easier by placing all known measurements into an equation that you have to work out like the ones written below. Below is a worksheet all about Chemical Reactions. On the front there are 8 listed statements and for each one I wrote yes if there was evidence of a chemical reaction. When I wrote no that meant there was no evidence of a chemical reaction.
On the bottom half of the worksheet I just filled in the blanks of the passage. It related to reactant's and product's. On the back of the page I took the Chemical Reaction given then wrote the balanced chemical equation. So what exactly is a Chemical Reaction? A chemical reaction is the change of a substance into a new one that has a different chemical identity. How can you tell if a reaction is happening? When there is a chemical reaction happening you can physically see changes. Example: The emission of heat and light, the formation of a precipitate, the evolution of gas, or a color change. But the only way you will know if a chemical reaction is really happening is validation with a chemical analysis of the products. There are 5 Types of Chemical Reactions. 1. A + B --> AB Synthesis- Two things becoming one 2.AB --> A + B Decomposition - A single compound undergoes a reaction that produces two or more simpler substances. 3. A + BC --> AC + B Single Replacement (Displacement) One element replaces a similar element in the compound. 4. AB + CD --> CB + AD Double Replacement- The ions of two compounds exchange places in an aqueous solution to form two new compounds. 5. Combustion- A substance combines with oxygen, releasing a large amount of energy in the form of light and heat. For organic compounds, such as hydrocarbons, the products of the combustion reaction are carbon dioxide and water. So there you have it, a brief summary all about Chemical Reaction. |
| Erica's Chemistry Blog |
|







 RSS Feed
RSS Feed